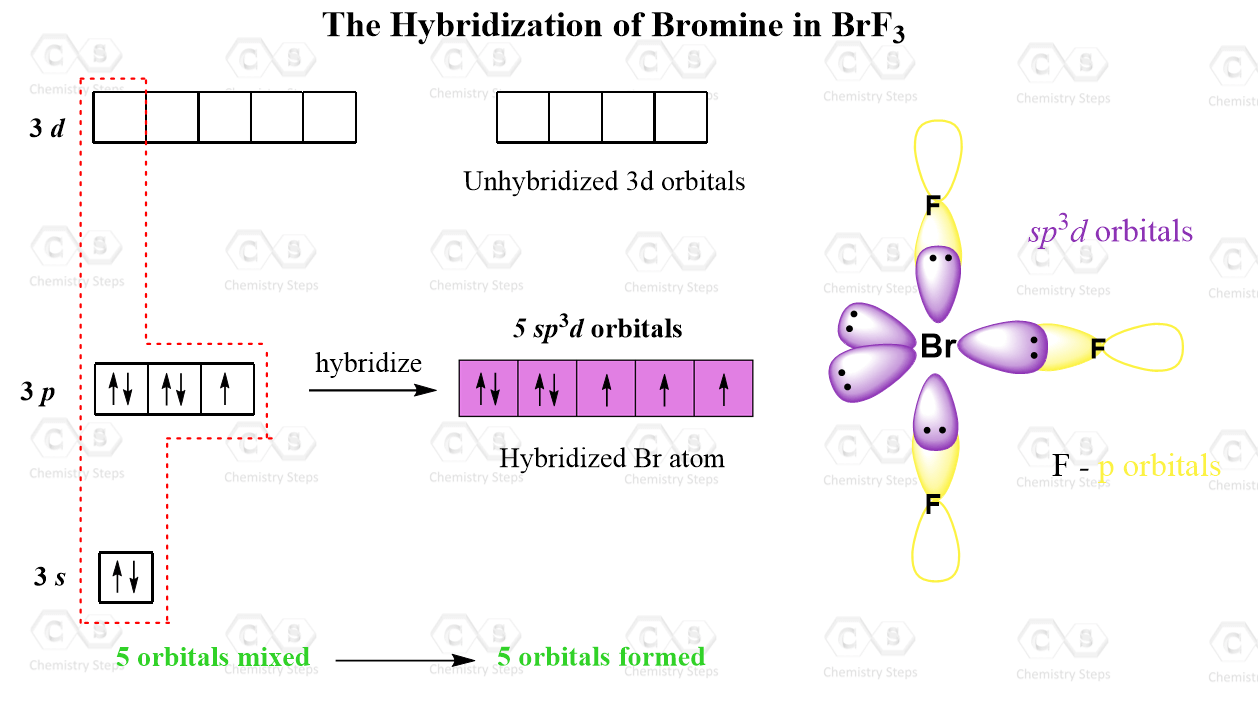
Hybridization of Atomic Orbitals
Introduction to the Hybridization Let’s start first by answering this question: Why do we need the hybridization theory? Here is one answer to this. It is confirmed experimentally that the carbon atom in methane (CH4) and other alkanes has a … Read more