 General Chemistry Cheat Sheets are PDF files written to be your reference guide for the key terms, concepts, and equations that may get buried in the overwhelming amount of material. The notes are not simply equations and formulas as they are accompanied by brief explanations, strategies, and some problem-solving examples. These study guides are beautifully crafted, powerful extract of information as a final touch before the test. General chemistry study guides will also help you prepare for AP, MCAT, PCAT, DAT, ACS, and other tests. The notes cover most of the chapters you will see in a typical undergraduate course. If purchased the bundle, you will receive the new guides as an e-mail as soon as they are published.
General Chemistry Cheat Sheets are PDF files written to be your reference guide for the key terms, concepts, and equations that may get buried in the overwhelming amount of material. The notes are not simply equations and formulas as they are accompanied by brief explanations, strategies, and some problem-solving examples. These study guides are beautifully crafted, powerful extract of information as a final touch before the test. General chemistry study guides will also help you prepare for AP, MCAT, PCAT, DAT, ACS, and other tests. The notes cover most of the chapters you will see in a typical undergraduate course. If purchased the bundle, you will receive the new guides as an e-mail as soon as they are published.
What is Included in Each Set?
General Chemistry Summary Guides Include
1. Significant Figures – 2 Pages

A summary study guide on understanding and counting significant figures. Rounding off to the correct number of significant figures, the rules for using significant figures in mathematical operations such as addition/subtraction and multiplication/division. The rules for significant figures expressed in scientific notation.
2. Atoms, Molecules, and Ions – 6 Pages

Atomic Structure, The Periodic Table, Names of Ions, Metals That Form Cations with Different Charges, Formulas and Names of Common Polyatomic Ions, Binary Compounds, Monoatomic Ions, Polyatomic Ions, Naming Ionic Compounds, Naming Hydrates, Naming Acids, Naming Bases, Formula from the Name. Worksheets on atomic particles, naming compounds, and writing formulas.
3. Mass, Mole, and Stoichiometry – 4 Pages

Atomic and Molecular Mass, Mole and Molar Mass, Calculating Moles from the Mass, Number of Atoms From Moles, Number of Atoms From the Mass, Number of Atoms in a Molecule, Percent Composition, Empirical Formula, Determining the Empirical and Molecular Formulas, Chemical Reactions and Chemical Equations, Balancing Chemical Equations, Indicating the States of Reactants and Products, Reaction Stoichiometry, Mass Calculations using Reaction Stoichiometry, Limiting Reactant/Reagent, Percent Yield
4. Reactions in Aqueous Solutions – 5 Pages

Solutions, The Solubility of Ionic Compounds, Electrolytes, and Nonelectrolytes, How to Write Dissociation Equations, Predicting the Products of a Reaction, Molecular, Ionic, and Net Ionic Equations, Solution Concentration, Concentration of Ions, Dilution, Stoichiometry of Reactions in Aqueous Solutions, Definitions of Acids and Bases, Acid–Base Titrations, Oxidation States, Rules for Assigning Oxidation Numbers, Determining Oxidation States, Oxidation-Reduction (Redox) Reactions, Balancing Redox Equations in Acidic and Basic Solutions
5. Gases – 2 Pages

Pressure, Pressure units, Gas Laws, Ideal Gas Law, Combined Gal Law and strategies to solve gas problems, Density and Molar mass of gases, Diffusion, Effusion, and Graham’ law for effusion and molar mass, Real Gases and the Van der Waals equation.
6. Thermochemistry – 2 Pages

Key Terms and Concepts in Thermochemistry, system, surroundings, internal energy, and its relation to heat and work, state functions, Enthalpy, exothermic and endothermic reactions, and their application in calculating the heat of reaction, Hess’s law, Heat Capacity, and Specific Heat, Specific heat capacity, Calorimetry, reactions under constant pressure, Bomb calorimetry, reaction when the volume is constant, Standard Enthalpies, Enthalpy of Reaction from Standard Enthalpies, Chemical Equations and Hrxn, Hess’s Law, Strategies an Examples of Problem-solving in Thermochemistry.
7. The Quantum-Mechanical Model of the Atom – 4 Pages
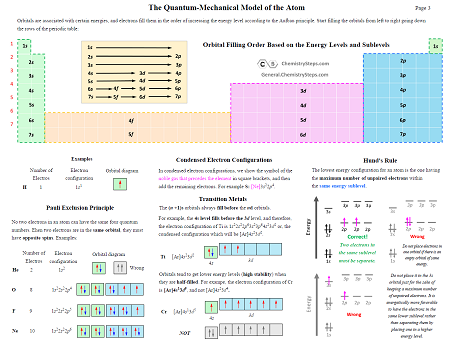
A summary study guide covering the key concept of the structure of atom such as the Bohr’s model of the hydrogen atom, the Photoelectric Effect, Electron Configurations, Quantum Numbers, Aufbaus’ Principle, Pailis’ Exclusion Principle, and HInd’s Rule with some worked examples.
8. Periodic Trends and Properties – 2 Pages

A summary study guide on the key trends and properties of the periodic table such as atomic and ionic radii, effective nuclear charge, ionization energy, electron affinity, and electronegativity.
9. Chemical Bonding – 7 Pages
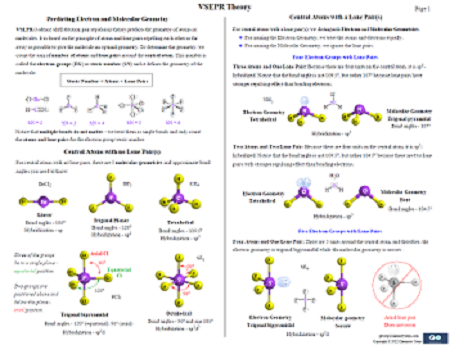
In this study guide, you will find the cleanest summary and graphics on valence electrons, Lewis structures, octet rule, formal charges, resonance structures, hybridization and VSEPR theories, sigma and pi bonds, polar, nonpolar, and ionic bonds, dipole moment, molecular dipole and more.
10. Colligative Properties – 2 Pages

Colligative (collective) properties – vapor pressure lowering, boiling point elevation (Tb), freezing point depression (Tf), and osmotic pressure (∏) together with example problems are summarized in this study guide
11. Chemical Kinetics – 5 Pages

Reaction Rate, Average Rate, Instantaneous Rate, Differential rate laws for the relationship of reaction and rate and concentration, Integrated Rate Laws for the relationship of concentration and time, Determining the rate laws using the initial rate method and graphing the data from integrated rate laws, Reaction Mechanism, Energy Diagram, Elementary Steps, Rate-Determining Step, Activation Energy, Catalysis, The Arrhenius Equation and some strategies and examples for problem-solving.
12. Chemical Equilibrium – 4 Pages

Reversible Reactions, Dynamic Equilibrium, Homogeneous and Heterogeneous equilibria, Equilibrium Constant (K), Key Facts About K, Equilibrium Constant and the Chemical Equation, Equilibrium Constants in Terms of Pressure, Kp, K for Heterogeneous Equilibria, The Direction of Reaction and Quotient (Q), ICE Tables, Le Châtelier’s Principle, How the equilibrium is affected when; the Concentrations are changed, The Pressure is changed, The Temperature is changed.
13. Thermodynamics – 5 Pages

Spontaneous and Nonspontaneous Processes, Thermodynamics vs Kinetics, Entropy and Spontaneity, Gibbs Free Energy, Correlating ΔG with ΔH and ΔS; how to determine if the reaction is spontaneous or not based on the enthalpy and entropy change, what role does temperature play in this, Predicting Entropy Change, Standard Entropy Change, Standard Free-Energy Changes, Free Energy change and the Hess’s Law, ΔG of a Reaction under Nonstandard Conditions, Relationship between ΔGo and Equilibrium Constant, The Temperature Dependence of K, Strategies, and Examples of Problem-solving in Thermodynamics
14. Acids and Bases – 7 Pages

Acid-Base theories, Strong and Weak Acids, Acid Strength and pKa, Factors that Affect the Acid Strength, Dissociation of Water, The pH Scale, Calculating the pH of a Strong Acid, The pOH Scale, Calculating the pH of Weak Acids, Weak Bases, and Base Ionization Constants, Calculating the pH of Weak Bases, Calculating the pH of Polyprotic Acids, Relationship Between Ka and Kb, Calculating the pH of Salts, Salts That Yield Acidic Solutions, Salts That Yield Basic Solutions, Salts That Contain Acidic Cations and Basic Anions, Buffer Solutions, How to Calculate the pH of a Buffer, The Common Ion Effect, The Henderson–Hasselbalch Equation, How to Prepare a Buffer with a Certain pH, Correlation of pKa and pH
15. Acid-Base Titrations and Titration Curves – 4 Pages
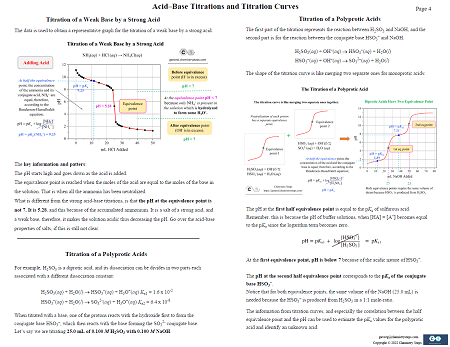
This study guide summarizes the key concepts of acid-base titrations, and titrations curves, for strong and weak acid-base pairs. Calculating the pH at different stages of the titration, equivalence point, half the equivalence point, the correlation of the pH and pKa, and the features specific to each type of titration.
16. Solubility Equilibria and Ksp – 3 Pages

The solubility product constant (Ksp), molar solubility, calculating solubility from Ksp and vice versa. The common ion effect, and the effect of pH on solubility with examples. How to determine if there will be a precipitate forming with a given concentration of ions.
17. Electrochemistry – 8 Pages
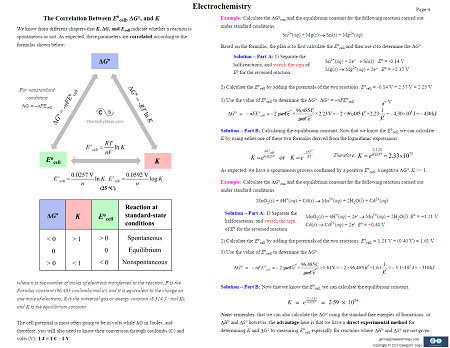
The Principles, Key Features, and Terminologies of Galvanic and Electrolytic Cells. Standard Electrode and Standard Reduction Potential – how to interpret it, some shortcuts for determining the stronger oxidizing or reducing agents, and the activity series of metals. Calculating the Cell Potential under Standard and Nonstandard Conditions. Nernst equation, Concentration Cells, Electrochemistry, and some more.
18. Nuclear Chemistry– 3 Pages

Review of Atomic Structure, Radioactivity and Nuclear Reactions, Transmutation, Alpha decay, Beta decay, Gamma radiation, Balancing Nuclear Equations, Positron Emission and Electron Capture, Nuclear Stability, Neutron to Proton Ratio and determining the type of nuclear reaction based on the band of stability, Magic Numbers, Rates of Radioactive Decay and Radiocarbon Dating, Energy Changes in Nuclear Reactions – why nuclear reactions are enormously exothermic, Nuclear Fission and Nuclear Fusion, Mass Defect and Nuclear Binding Energy with example calculations.
Get the bundle of all General Chemistry Study Guides
$17 Bundle
Detailed Beautiful Powerful
The General Chemistry Study Guides come after four years since the organic chemistry notes were released and thousands of copies were sold. And if you never used or heard about them, check the reviews below and leave yours.
Excellent explanation with graphs
It is wonderfull to work with you
Answers
great
It is very helpful ☺️
Add orbitals energy levels diagram to study guide 07 – The Quantum-Mechanical Model of the Atom
good
Everything about this channel is very smart. It simply shows exercises and saves time and energy.
It was nice though because it make me learn and it has also a question and answer
Combined Gases
Boyles’s Law
Charles’ Law
Gay Lussac’s Law
vapor pressure
So nice at all ..it has made chemistry to be easier to me ❤️
Solubility Equilibria and Ksp: Can you please add complex ion notes
Student
Okayed
Quiz
Outstanding
This has been so helpful
This is the best site have come across to so far.
Response from Chemistry Steps
Glad to be helpful, Danny. Thank you!
Very helpful
very very useful. Thanks
The way you break down the topics in these study guides is simply amazing. A huge help in helping me pass chem 2 this summer.
excellent!
5
Very useful website for JEE-Advanced
Very Useful website. This is really great work. I saved time and money. Thanks.
Response from Chemistry Steps
Glad to have you joining, Roman.
Outstanding website, the best so far, and I strongly recommended it to my students! You have done an excellent job in providing clear, step-by-step, color coded, easy to understand guides, and I believe your next step should be a textbook! Just excellent!
Response from Chemistry Steps
Thank you very much, Asmik!
I have been using Chemistry Steps for Organic 1 and 2 and struggled in the first semester before finding this site and I learned here more than in the zoom lectures and the book. The types of practice problems and the solutions are extremely relevant, and the addition of new puzzles is what I was looking for since my professors likes to give tons of synthesis. It’s great – highly recommended. The only regret is not knowing about it early enough in the O Chem 1 semester.
Response from Chemistry Steps
Great to hear it’s helping you, Gabi.
Fabulous and valuable source to guide u in branches of chemisty. But l gonna to register for free …..
Very interesting website.
I like it. Thnx
Amazing
Very good
For Mphill admission test at any university i found good content
Excellent
good
Very good. It is really needed for the one who wants to move ahead
Very good
Teacher of organic chemisty. Researcher in green cehmistry
very good content
I want to study organic chemistry
Wow
Good content with numerous examples
Excellent content
Great for org chem!
It’s very good and i’ve learned a lot from it
Very good
I love that your posts and summaries are short and clear to the point. There is too much fluff in organic chemistry confusing us and making things complicated.
good
It is valuable material. If you have any possibility please send these to my mail as below. Thnq
very helpfull
Excellent, in-depth study material! Every little detail related to the topic is there and concepts are summarized in worked examples.
G. Paoletti
Going from Org I to Org II was such a rough turn. I did well in Org I but had almost no hope of passing the Org II taking in the summer semester. I learned so much in one week form these cheat sheets and I actually passed the class with ease. I only wish I would have bought these at the beginning of the semester and I could have done better.
Briana Paisley
I wanted to thank you so much. Unbelievable how much information is found in one study guide summary sheet and how much I did not need from my textbook.
Jeffery
This is great work. I can’t fathom spending this much time on something, after a peruse it genuinely looks very useful. I just can’t believe I haven’t heard of it sooner. It’s likely I will purchase a prime membership soon and I wholly think it’s going to be a difference maker in my degree. Thanks a lot for your work. More people need to know about this!
Deikon
I came across these accidentally and found my rescue in the battle with organic chemistry. They cover everything about the alkene, alkyne, aromatic and all those countless numbers of reactions. And what is amazing is that I am able to read and learn as these are not just cheat sheets where everything is crammed in one place. It is like reading a book with a much better focus getting straight to the point in an easy to follow structure. Additionally, after going over the topic, there is plenty of practice accessible on a click. Yes, the answers are paid but they are worth it. I jumped from a 72 on my second exam to a 96 on my last exam. That is if you are in doubt about whether to buy these fantastic study guides or not.
Sylvia Lawrence
These study guides are extremely helpful when it gets so hard to keep up with organic chemistry lectures. Great summaries to master and solidify the key concepts right before the test. I highly recommend them.
Greg
“Sir, I owe you my grade. Yes, I was impressed by these detailed study guides right when I downloaded them, but I am e-mailing you now that the brutal semester is over and I got an A in a class that I thought I’d never be able to handle.“
Alyssa Belcher
You cannot change the nature of the subject, but these study guides ease the battle with organic chemistry.
Sam Royle
Watching videos wasn’t enough to grasp the concepts I was searching places to practice orgo. So may exercises here and adding quizzes was definitely a great help. Better than my textbook without the expensive solutions manual.
Angie
Unfortunately, I came across this site a little too late in my Organic Chemistry 1 course. Shame… Didn’t leave it till the last minute before the final. Super helpful quizzes and study guides. Thanks for the finals discount!
Alexandra
Very informative, your site got me through the semester. More people should know about it.
Erika
The most comprehensive study guides. I wouldn’t survive all the reactions of the carboxylic acid derivatives.
Synthia Hudson – Penn State
Thanks so much for that wonderful site you have! I currently teach Chemistry II at a high school in Alabama. This class is mostly organic chemistry. I have a subscription to your site and use the resources to help me prepare my lessons.
E. Harris
Thank you! Your website helped me a lot this semester when I didn’t get to see my professor for the most part.
Alexandra at CSU
I am very excited to study and understand organic chemistry more.
Yasser
It is a fantastic website for easing organic chemistry.
Omarkemia
Thank you for your web site!!!! It gives an excellent understanding of how Organic Chemistry ”works”. Very good exercises!!! Solving these problems gives the opportunity to understand how good you are in Organic Chemistry.
N. Durgaryan
I loved the website.
Lavanya
I love this site and l want to join it
Maysam khateeb
The best page of Chemistry
Carlos Molina
Your articles are very very helpful sir
Sandeep Prajapati
Used it throughout the semester and the practice problems helped me a lot with the tests. Definitely recommended!
Victoria Norris
Ottimo sito.
Leonardo Rapisarda
Your exercises are helpful.
Ariana R
This website is the best option if you want to ease your life during Chemistry classes and be prepared for your exams. I thoroughly recommend joining them
Arturs Malusickis
The cheat sheet is beautifully designed and easy to follow.
Alexander Rorong
The notes are very organized and helpful.
Kathy Phan
This website is awesome
Jordan Howery
It’s really good to be here
Agness S Kamara
Very helpful indeed. Organized! That’s what l was looking for.
Charity
What an amazing and comprehensive summary, I am grateful for coming across this vital write up, I enjoy my glancing through this script.
Al-katangi
After a secure and quick checkout (no name, no phone, no address), you will receive a direct link and an e-mail to download all the summary sheets as high-resolution searchable PDF files.
The file can be downloaded one-by-one or all at once by scrolling to the bottom of the page.
General Chemistry 1 and 2 Study Guides
Get everything in one place!
The transactions are processed by Stripe and PayPayl gateways with all the security and privacy policies given the highest importance and priority.


