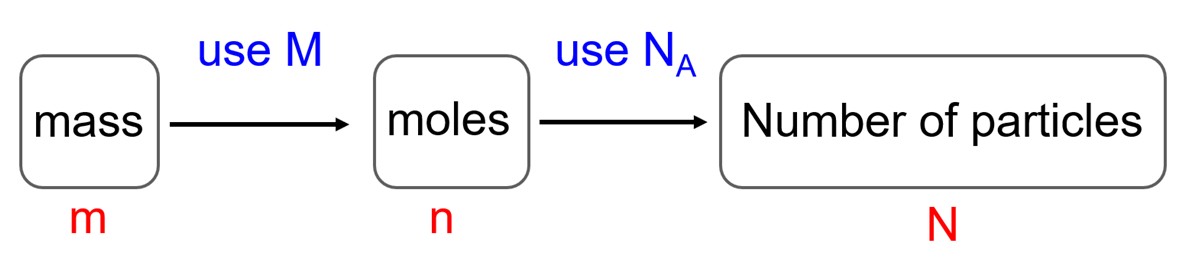
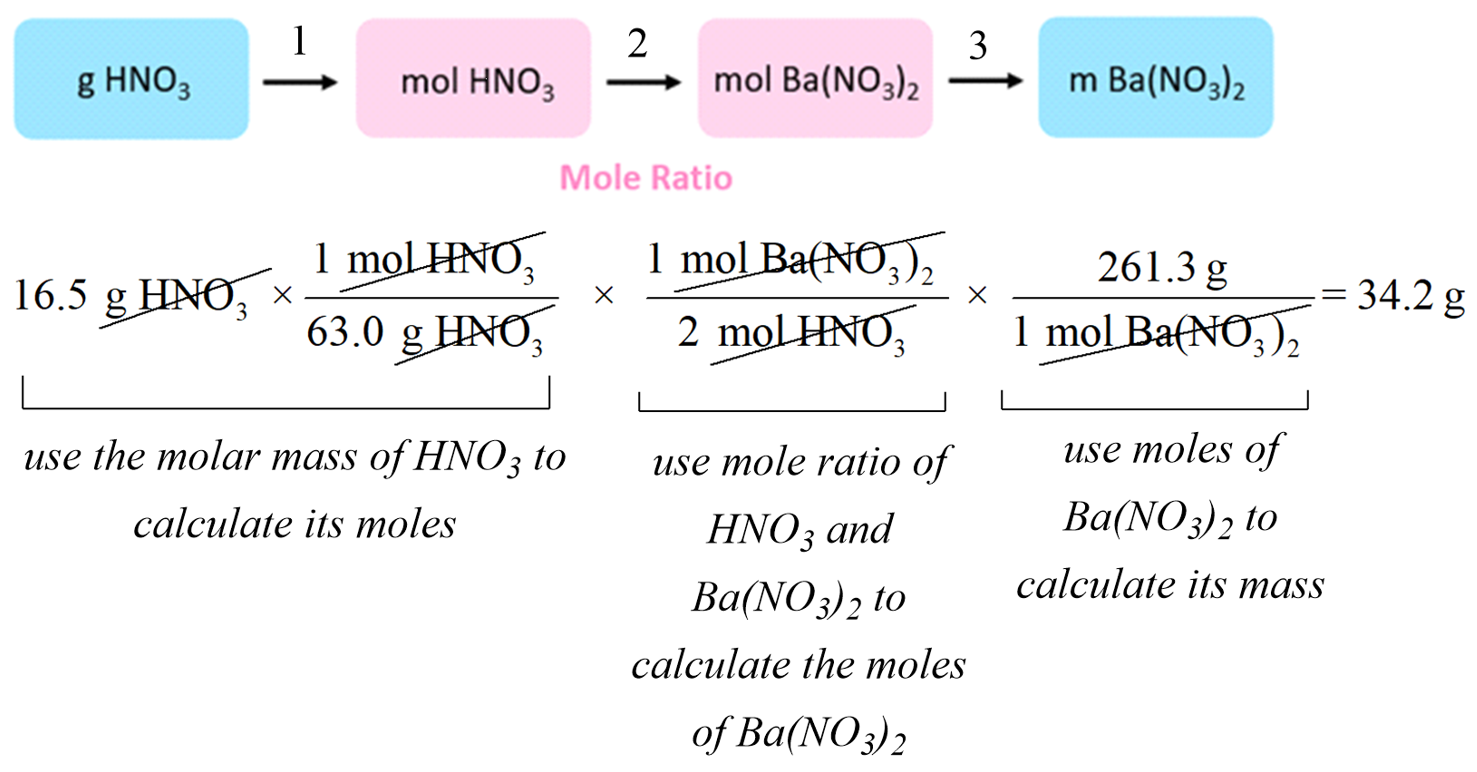
How To Find The Amount of Excess Reactant
When two substances are reacted in a nonstoichiometric ratio, one of them is going to be the limiting reactant (LR), and the other is the excess reactant. The limiting reactant is the one that runs out first, and therefore, defines … Read more