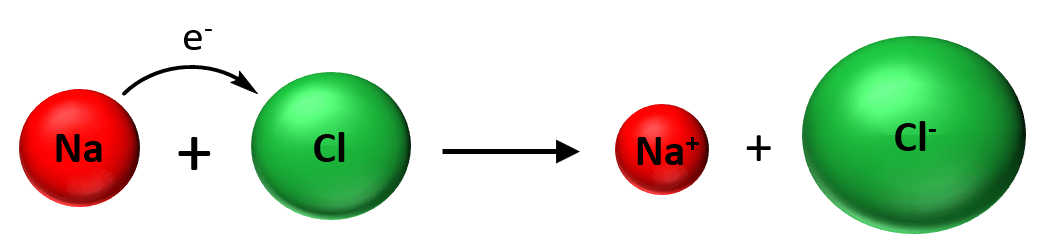
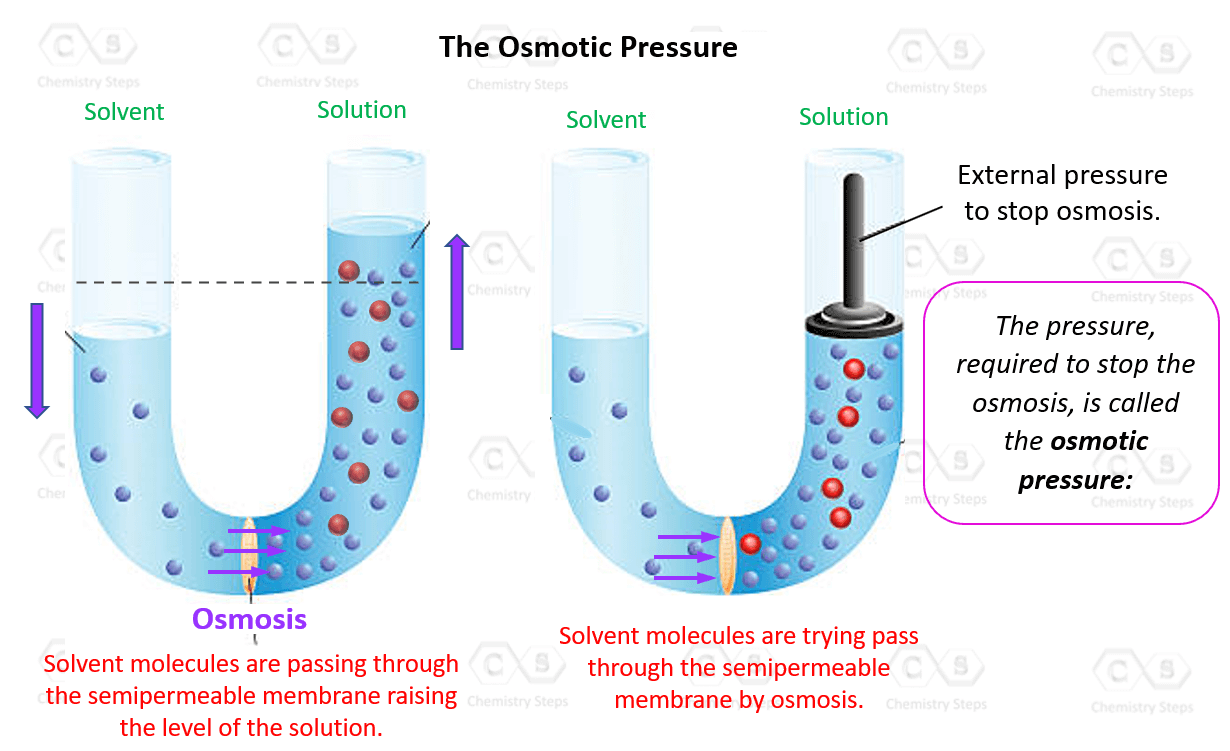
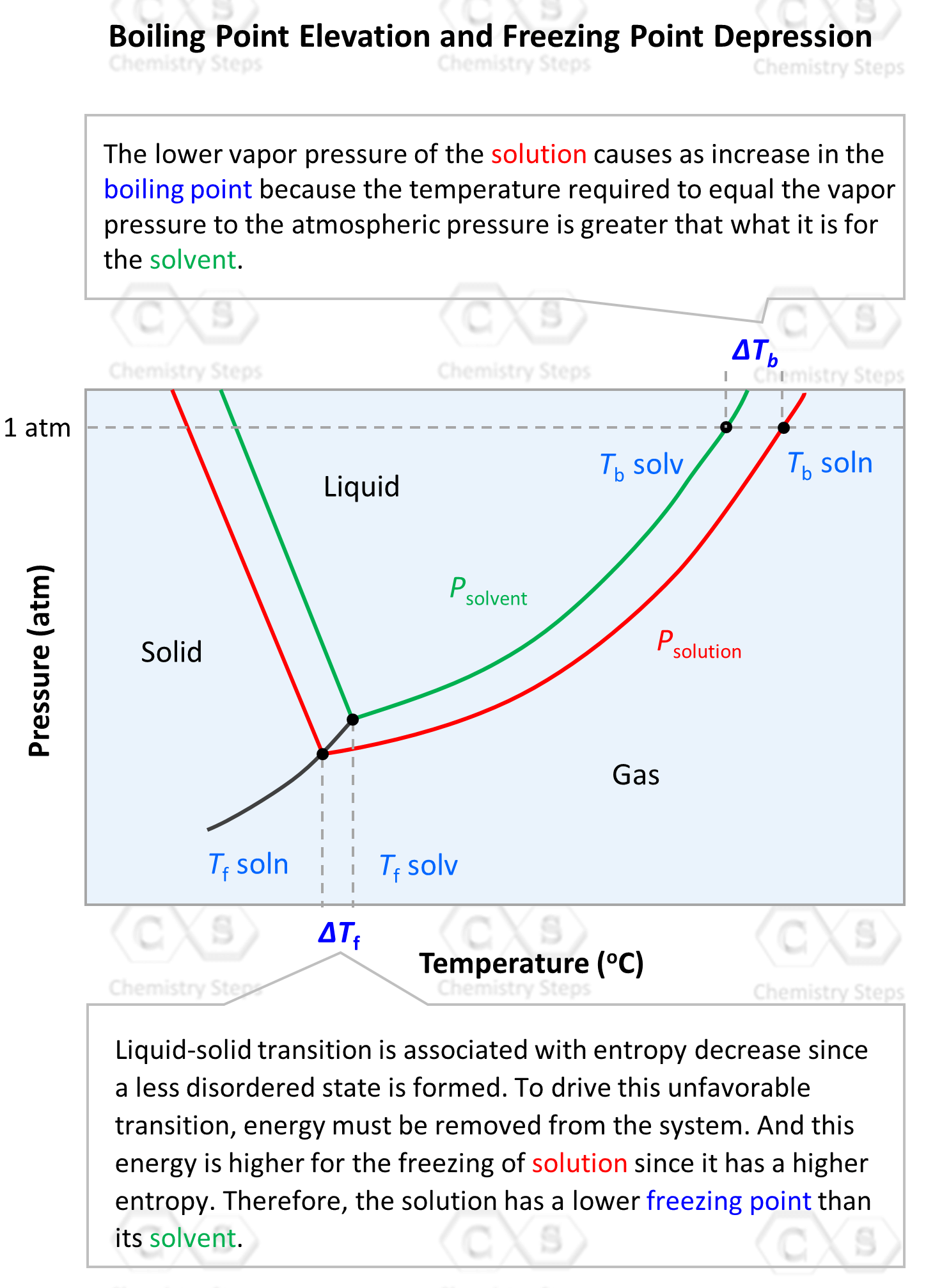
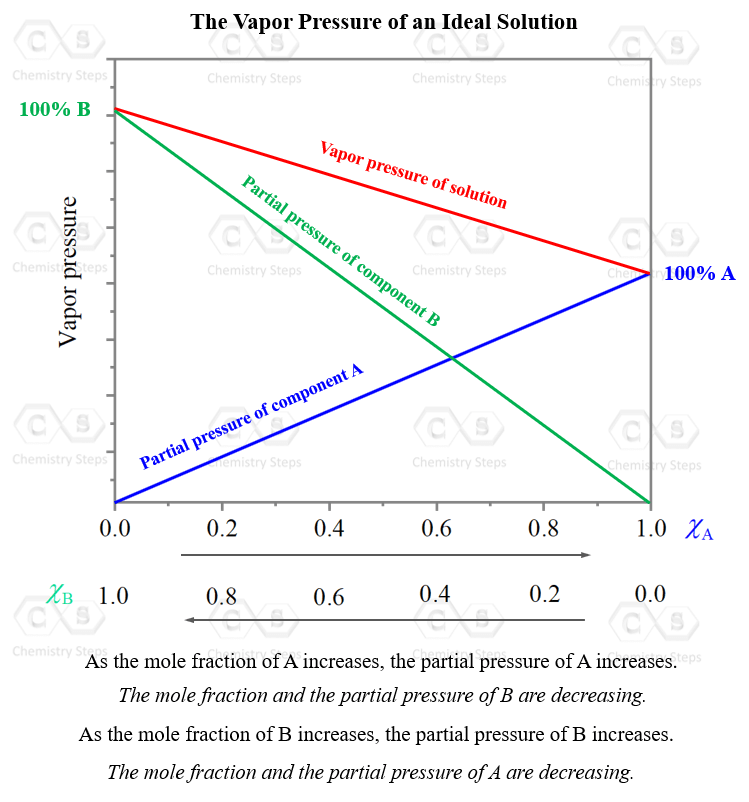
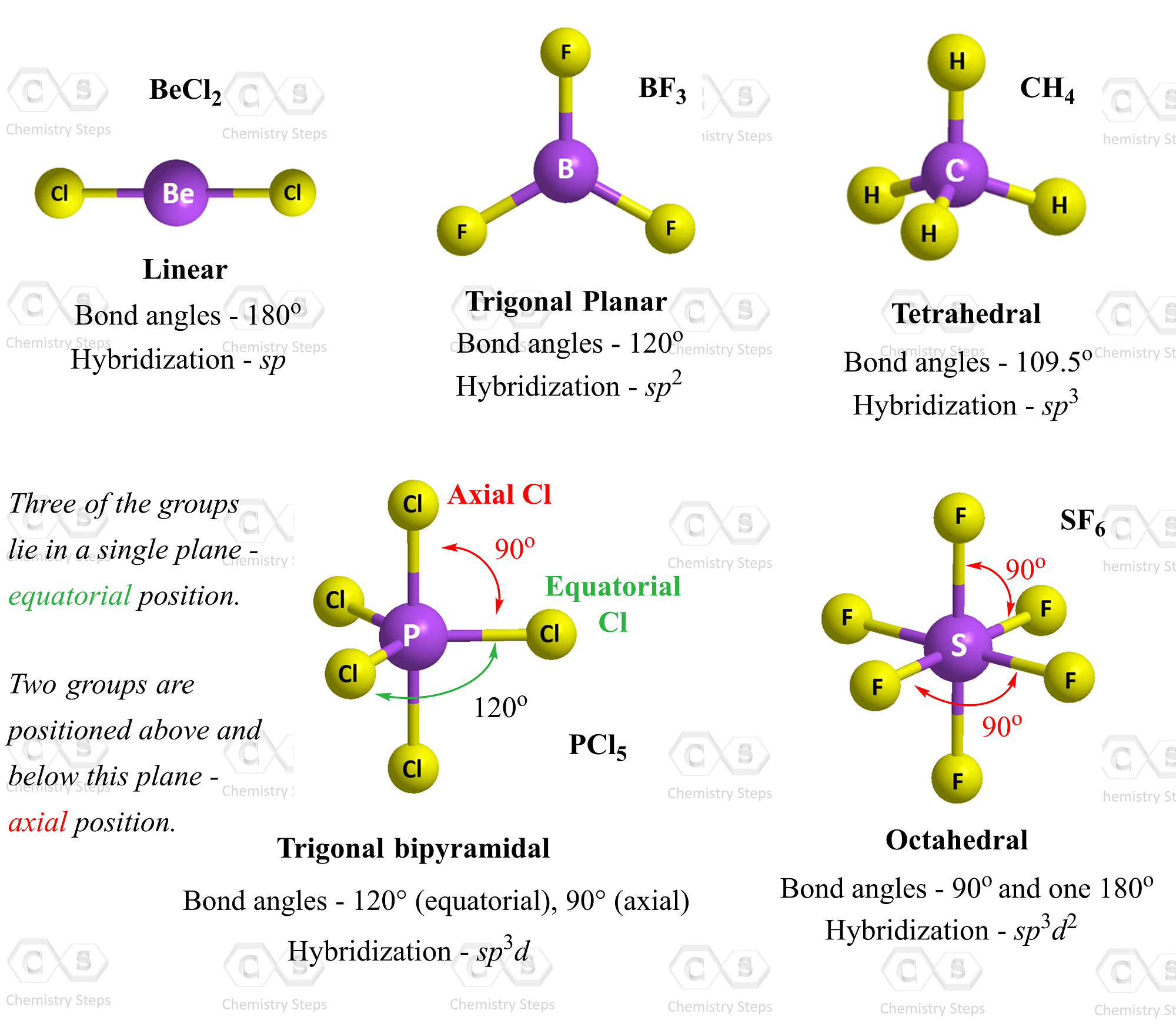
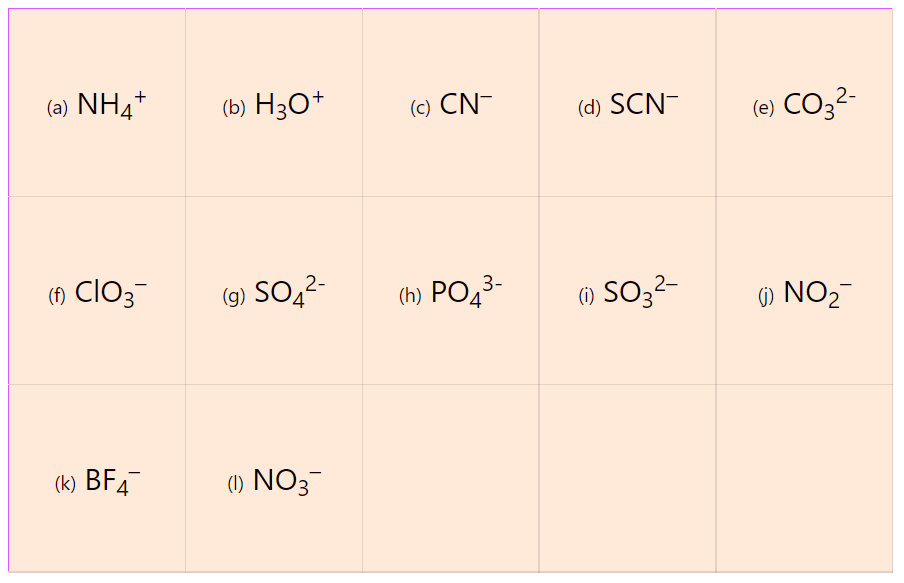
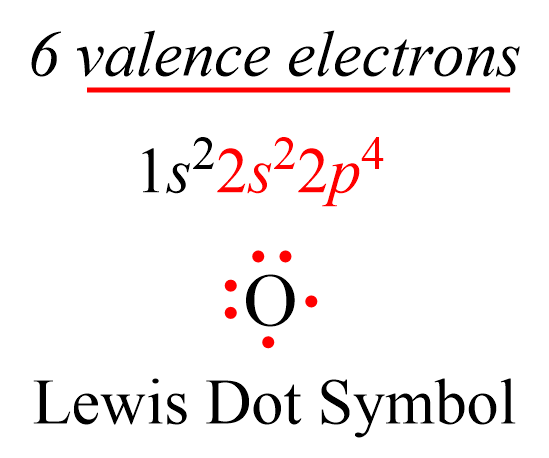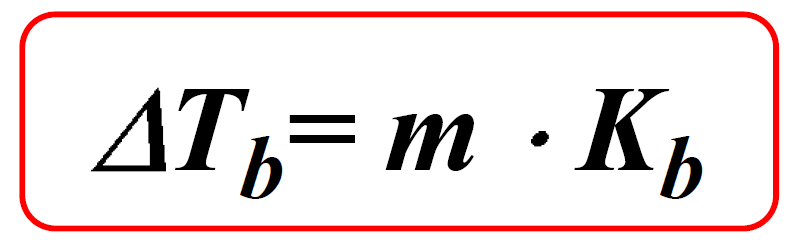Ionic Bond
The ionic bond is normally formed between metals and nonmetals and the reason for this is their different electron affinity. Remember, metals have low ionization energies and therefore, tend to form cations, while nonmetals have high electron affinity and tend … Read more