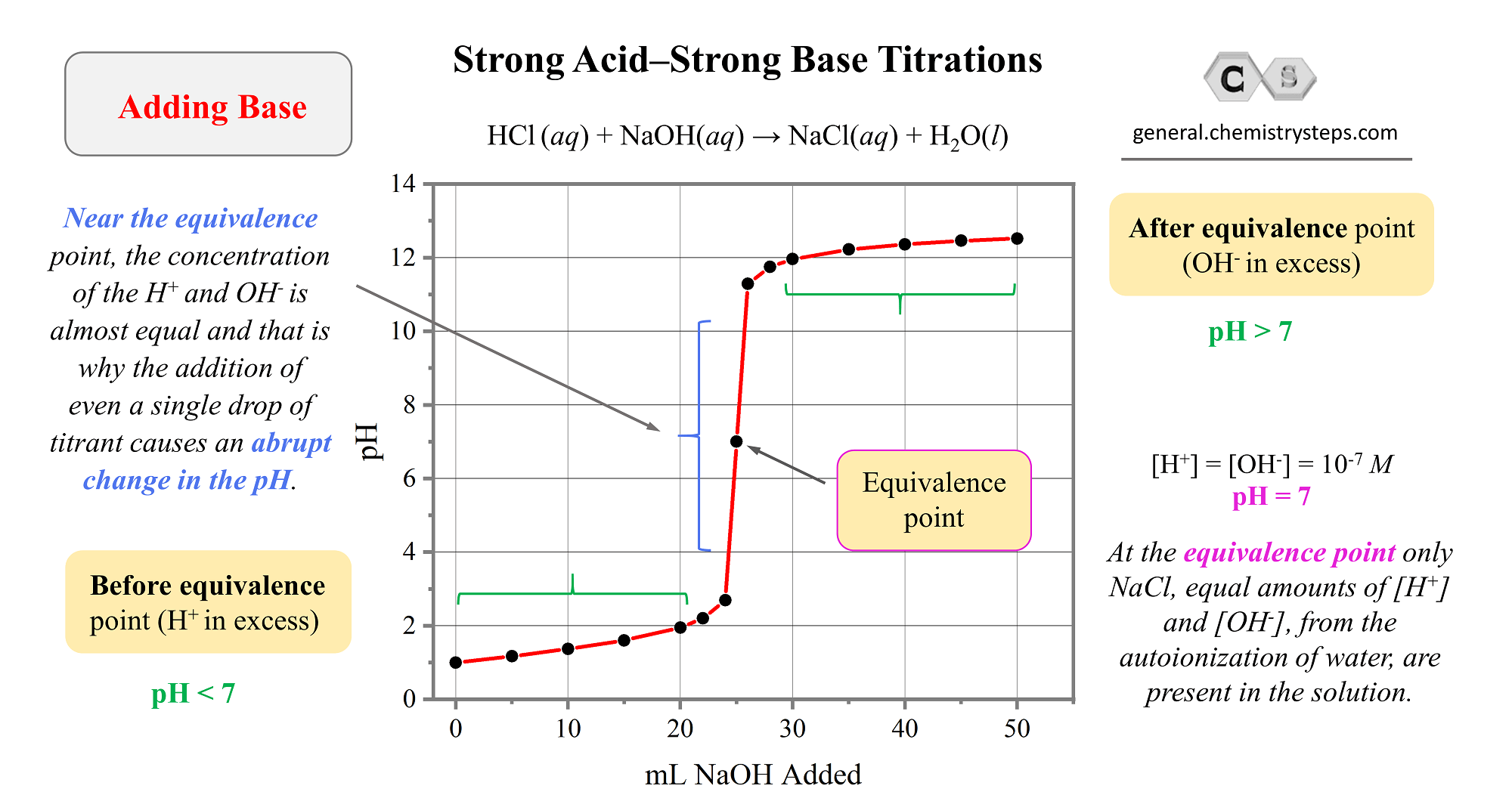
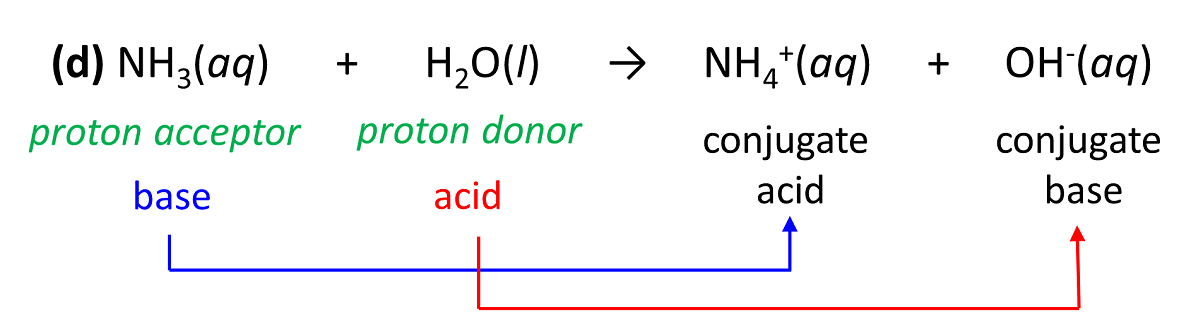
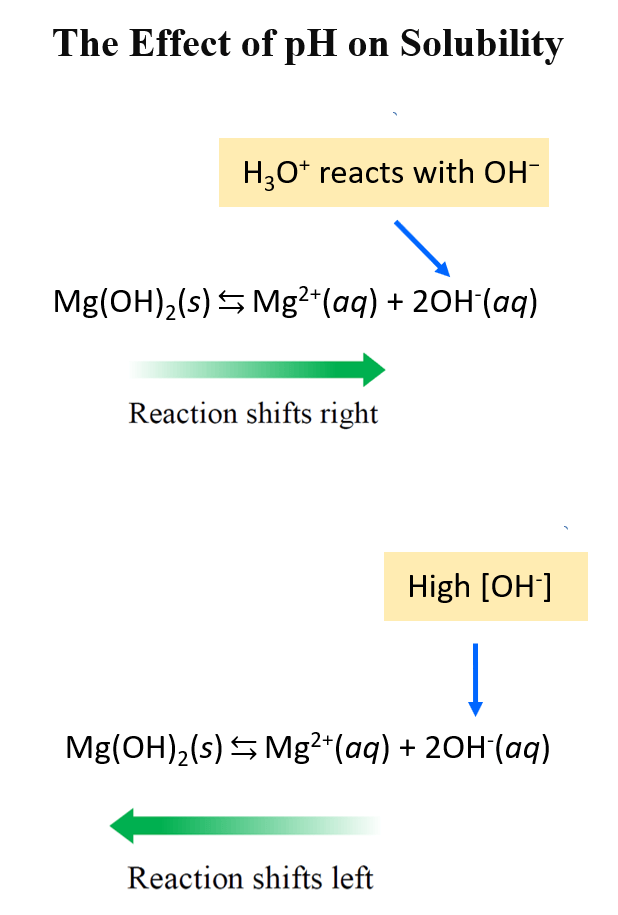
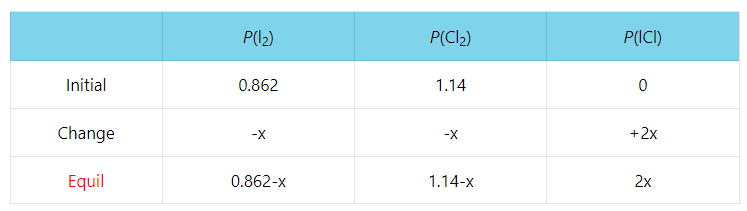
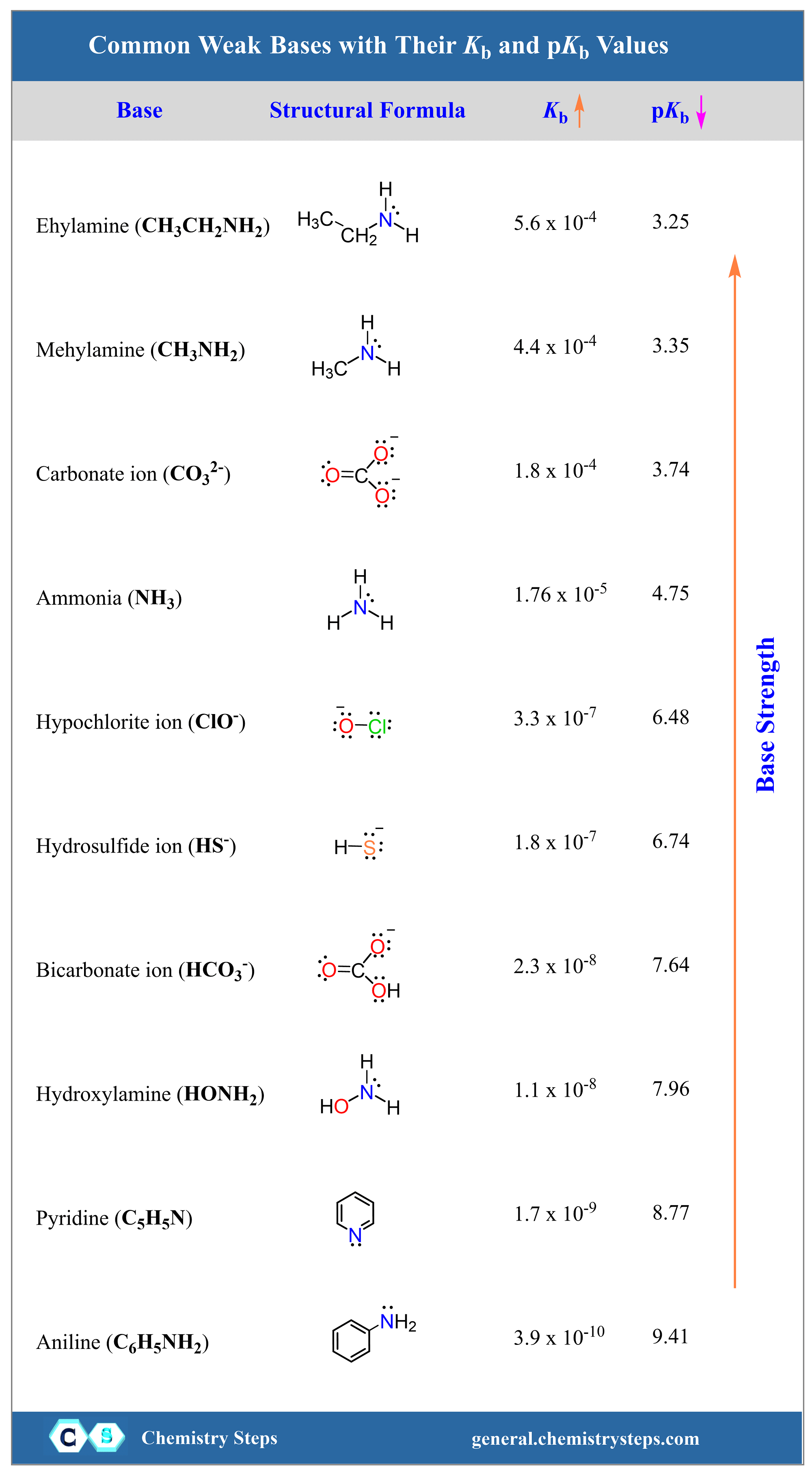
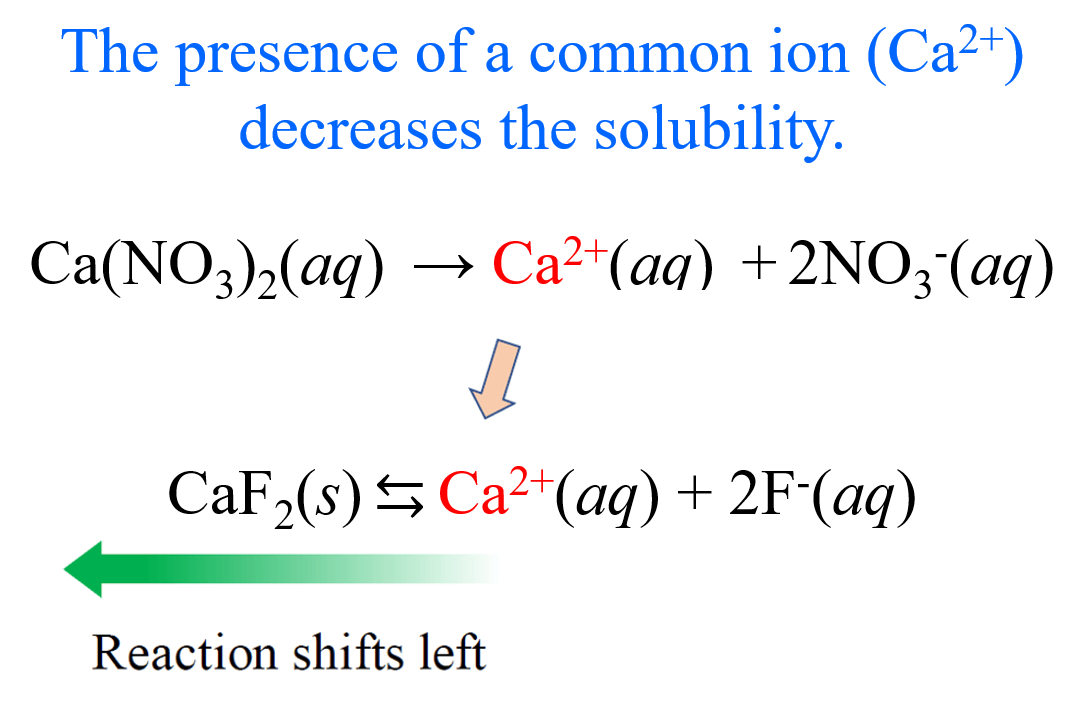Titration of a Weak Acid by a Strong Base
In the previous post, we discussed the titration of 25.0 mL of 0.100 M strong acid HCl with 0.100 M NaOH. Today, we will focus on the titration of a weak acid with strong bases, so we can still use … Read more