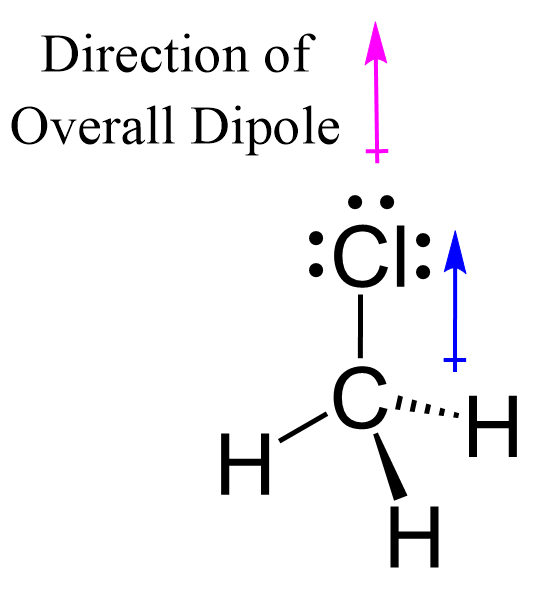
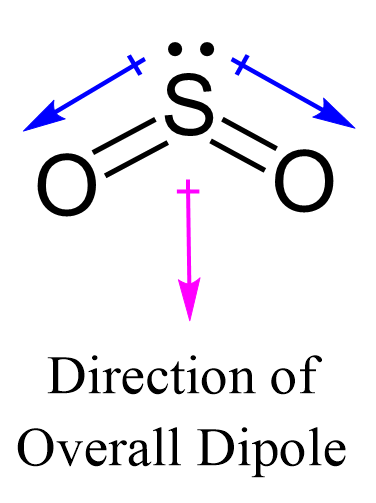
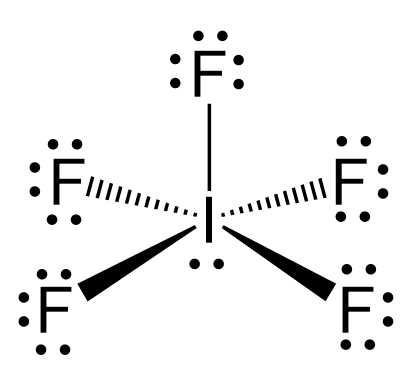
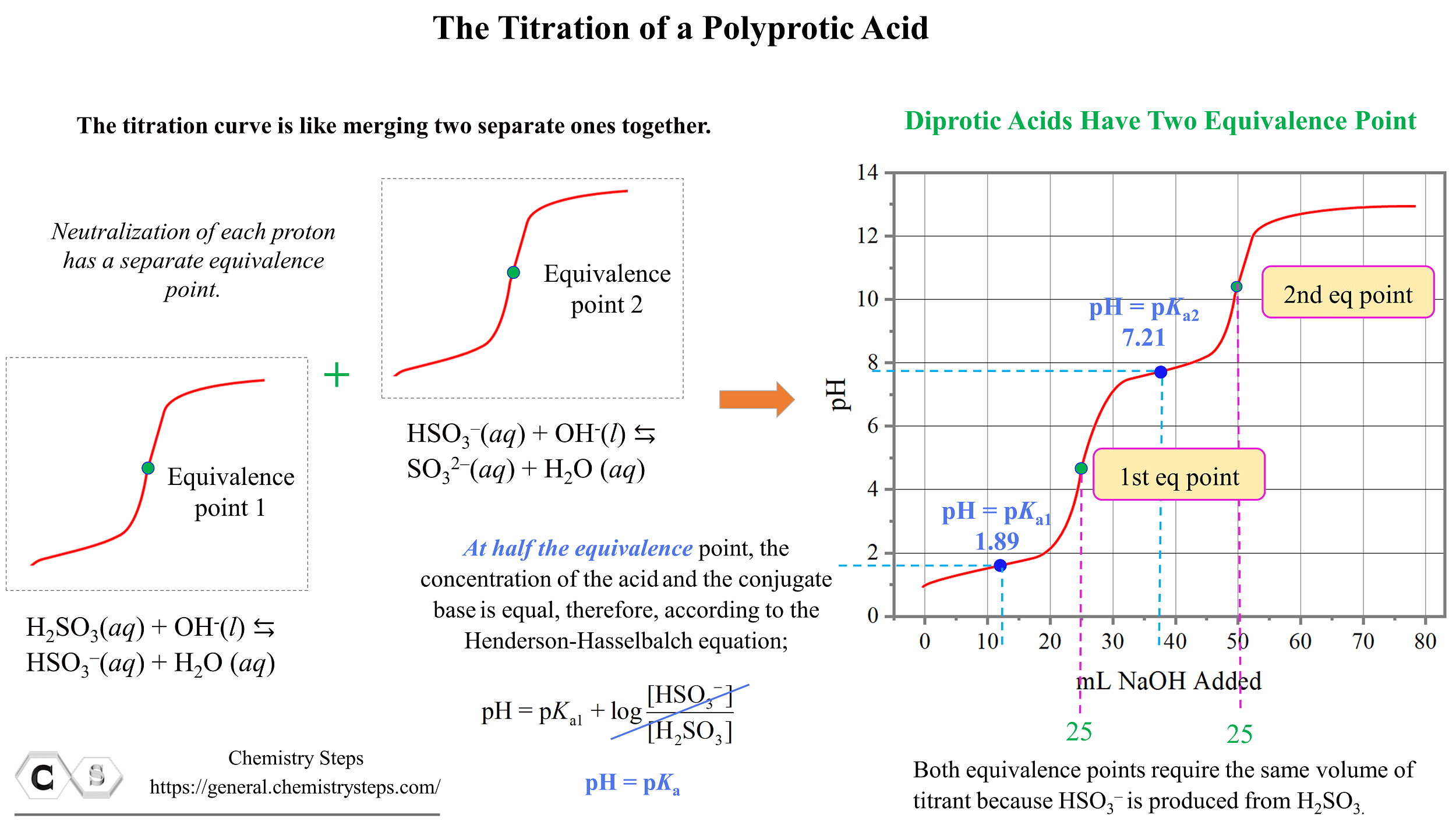
P4O10 Oxidation Numbers
To determine the oxidation number of an atom(s) in a molecule or an ion, start with the known oxidation numbers and the rules summarized below: Keep in mind that the summary is zero for neutral molecules and is equal to … Read more