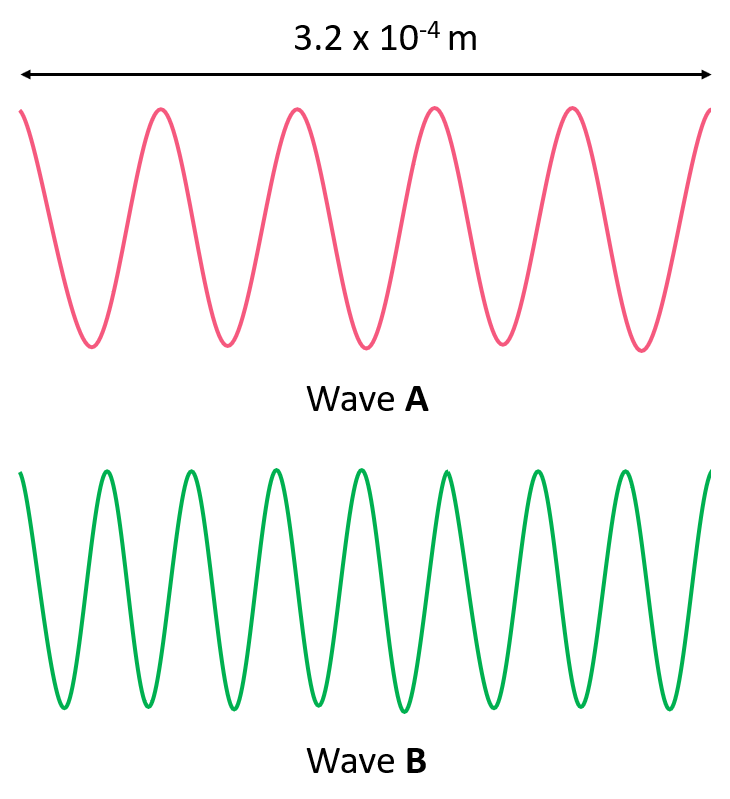
Energy, Wavelength, and Frequency Practice Problems
In these practice problems, we will go over examples of determining the wavelength, frequency, and energy of light, calculating the number of photons in a laser pulse based on the energy, understanding the correlation between the energy and wavelength and … Read more