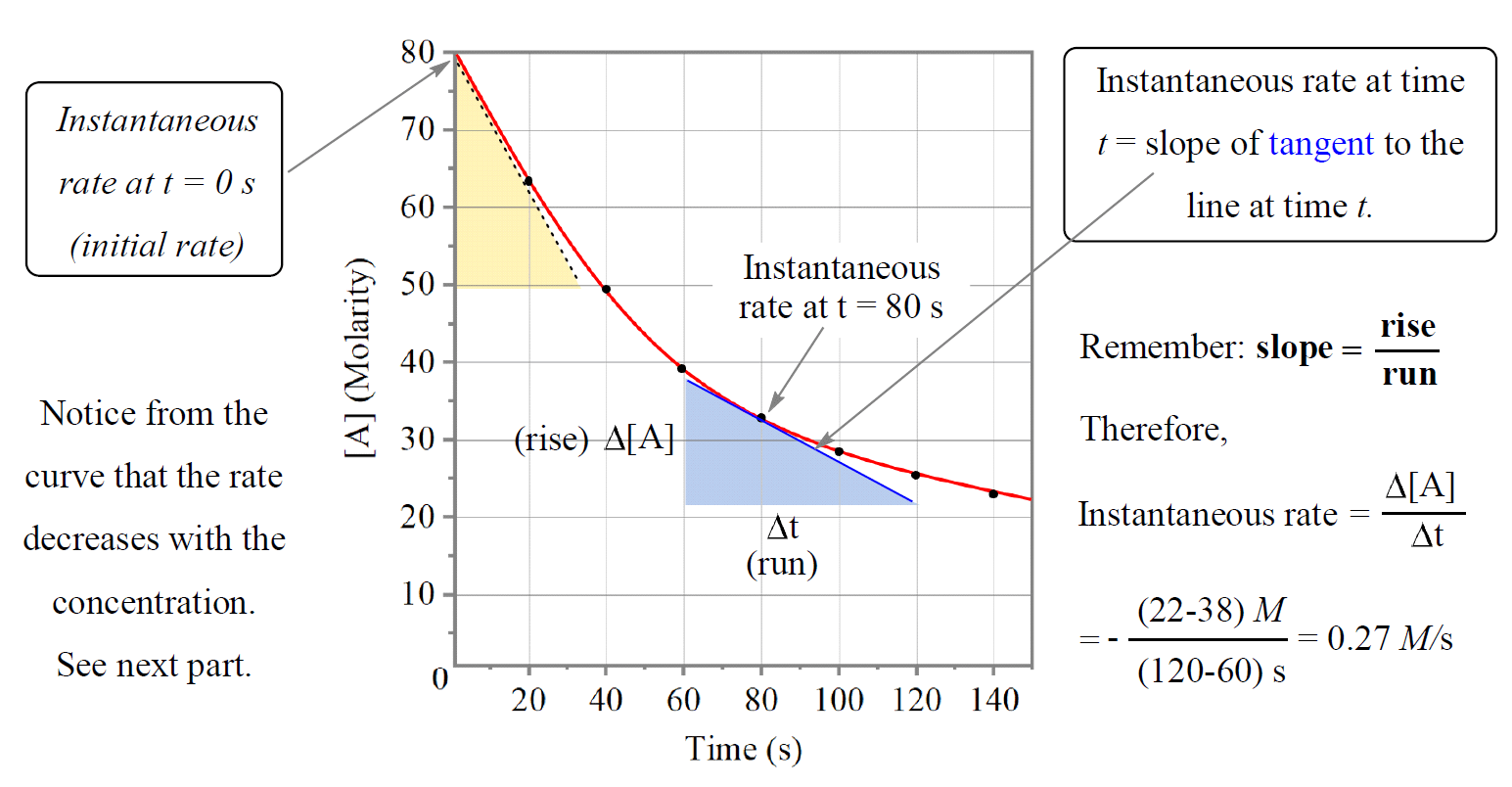
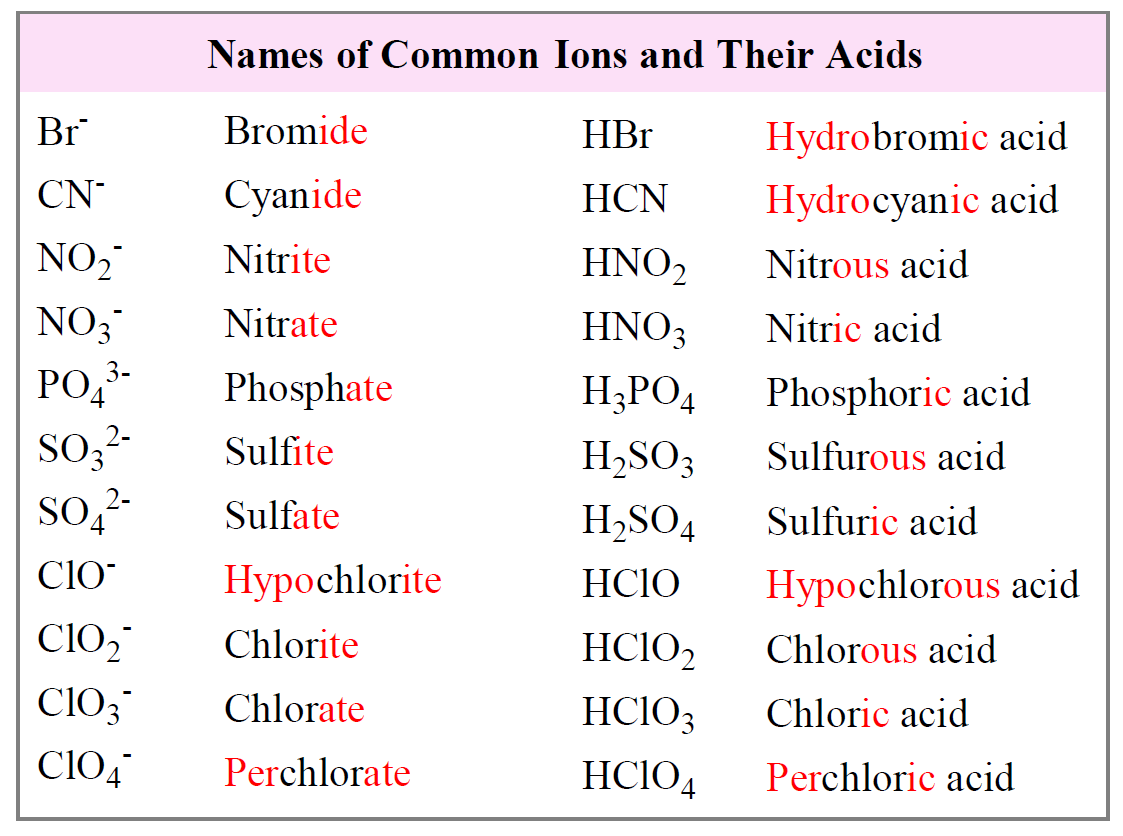
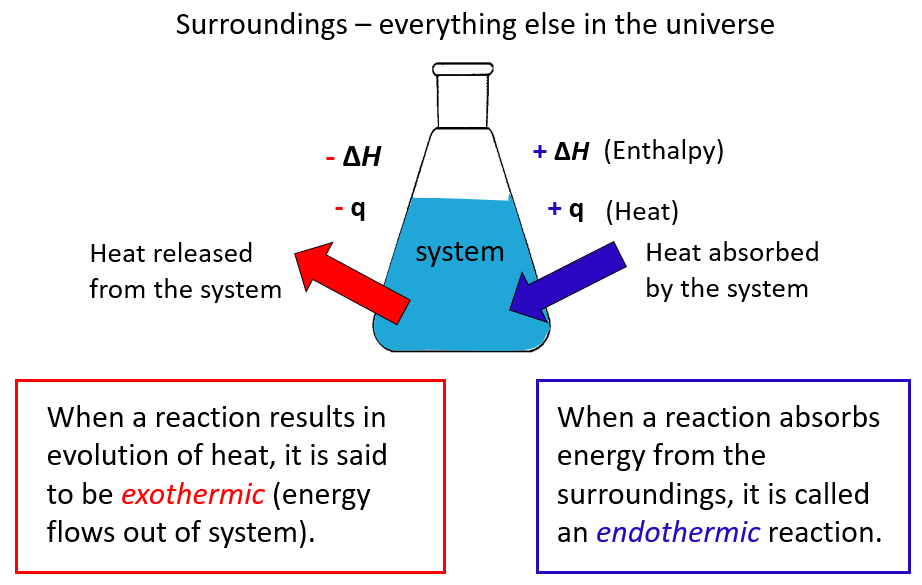
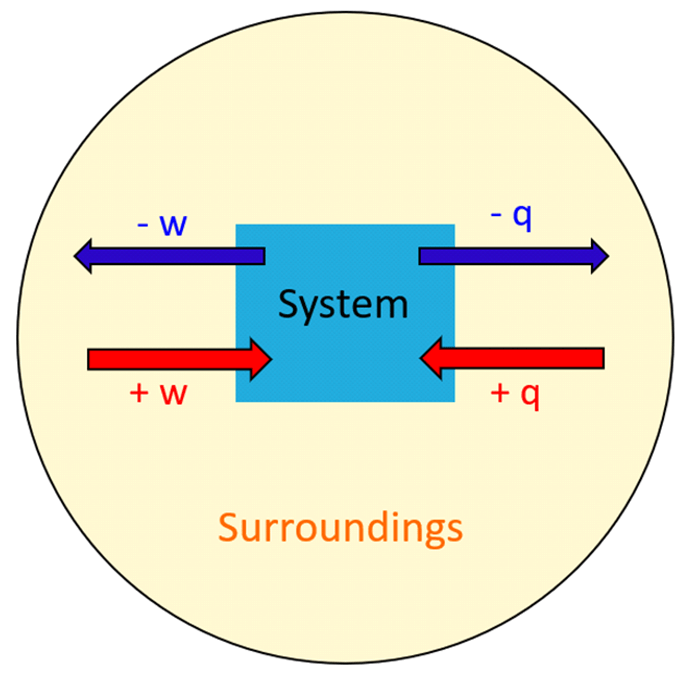
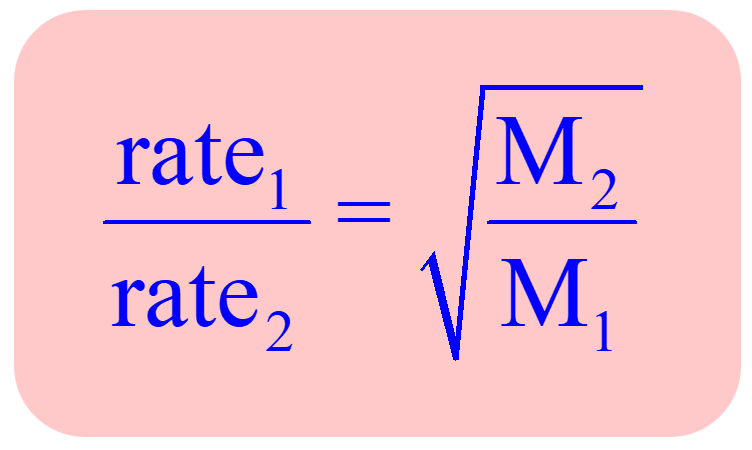Rate Law and Reaction Order
In the previous post, we talked about the reaction rate and saw that it is not constant and changes with the concentration of reactants. This is not true for all reactions and it’s possible that the rate may be independent … Read more