To determine if CBr4 is polar or nonpolar, we need to first determine its geometry. This presumes knowing the rules for drawing a correct Lewis structure and you can find more details about Lewis structures here.
Carbon is the central atom, so we can draw the skeletal structure:
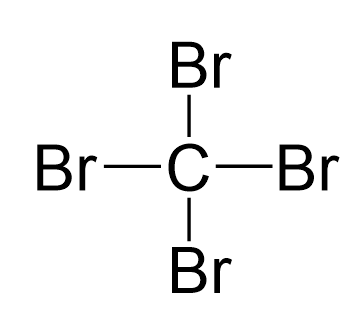
There are 4 + 4×7 = 32 electrons, and 8 have been used to make four bonds. The remaining 24 go on the two bromines as lone pairs:
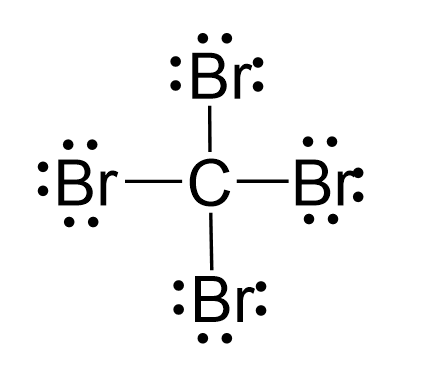
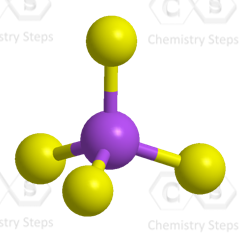
The central atom has four atoms and no lone pair, therefore, both the electron and molecular geometries are tetrahedral:
Now, the polarity: The first thing here is to determine if the C-Br bond is polar. Depending on the difference in the electronegativity values, covalent bonds can be polar and nonpolar.

- If the difference in electronegativity is less than 0.5, the electrons are about equally shared between the two atoms, forming a nonpolar a covalent bond.
- If the difference in electronegativity is between 0.5 and 1.7, we have a polar covalent bond.
- A difference of 1.7 or higher is so large that the electrons are no longer shared, and an ionic bond is formed. Ionic bonds are formed between metals and nonmetals.
Even though the C-Br bond is considered polar, the symmetrical shape of the molecule results in no molecular polarity.
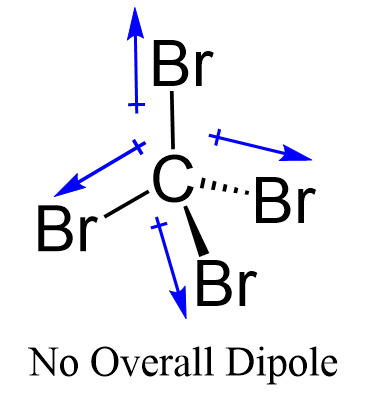
Remember, the net dipole of the molecule is the vector sum of all the dipoles and here it equals to zero because the bonds are equivalent and the four dipole moments are pointing in opposite directions.
You may be wondering why the difference of 0.30 makes the C-Br bond polar. One explanation can be the larger size and thus the polarizability of the bromine. Also, we know from the certain reactivities of the C-Br bond in organic chemistry that it has a polar nature.
Check this 99-question multiple-choice quiz on Geometry and Hybridization:
Geometry and Hybridization Quiz
Check Also
- The VSEPR Model
- VSEPR Theory Practice Problems
- Hybridization of Atomic Orbitals
- sp, sp2, sp3, sp3d, and sp3d2 Hybridization Practice Problems

