Mole Fraction and Partial Pressure of the Gas
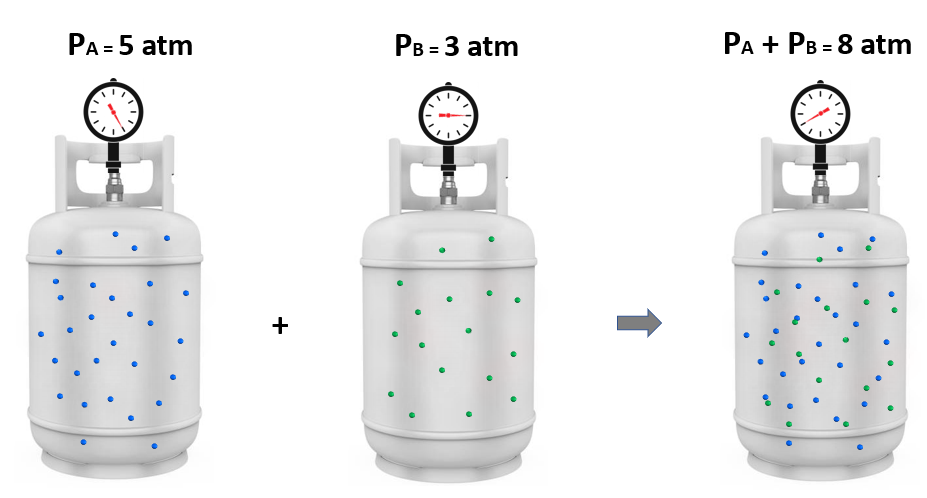
In the previous post, we talked about partial pressures and the Dalton law. Today, we will see how the mole fraction and partial pressures of a gas are related. Remember, mole fraction (Χ) is the ratio of the moles of … Read more