Sulfur is the central atom:
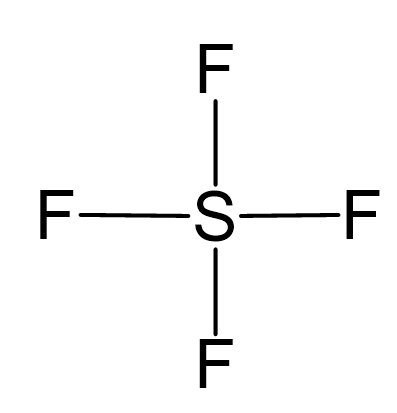
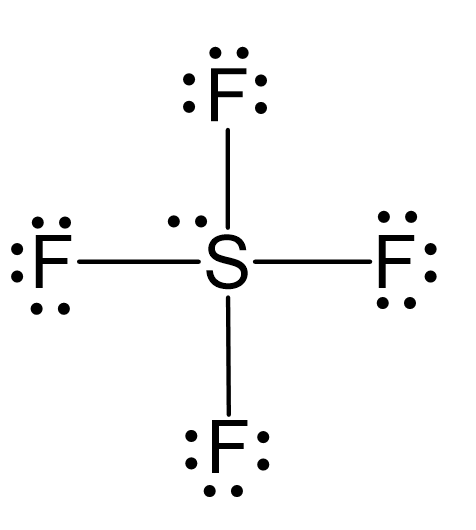
There are 6 + 4×7 = 34 valence electrons, and 8 are taken for making 4 covalent bonds. Each fluorine takes three lone pairs, so there are 34 – (8 + 4×6) = 2 electrons left which go to the sulfur as a lone pair:
The central atom has 4 atoms connected to it, and one lone pair, therefore, the electron geometry is trigonal bipyramidal while the molecular geometry is seesaw:
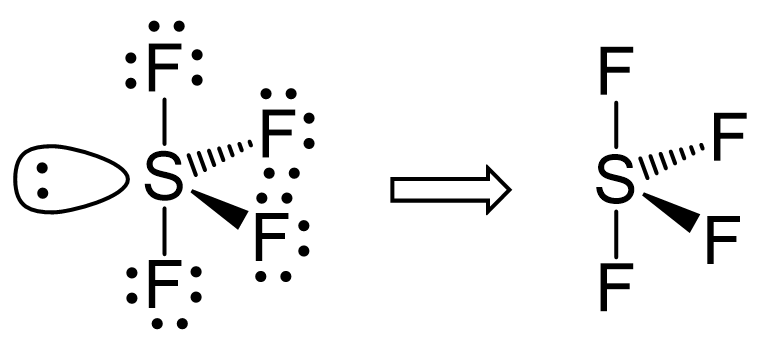
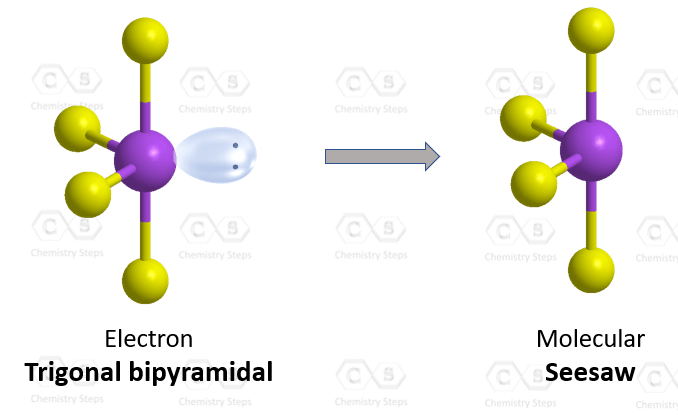
Notice that the lone pair does not go in an axial position (up or down).
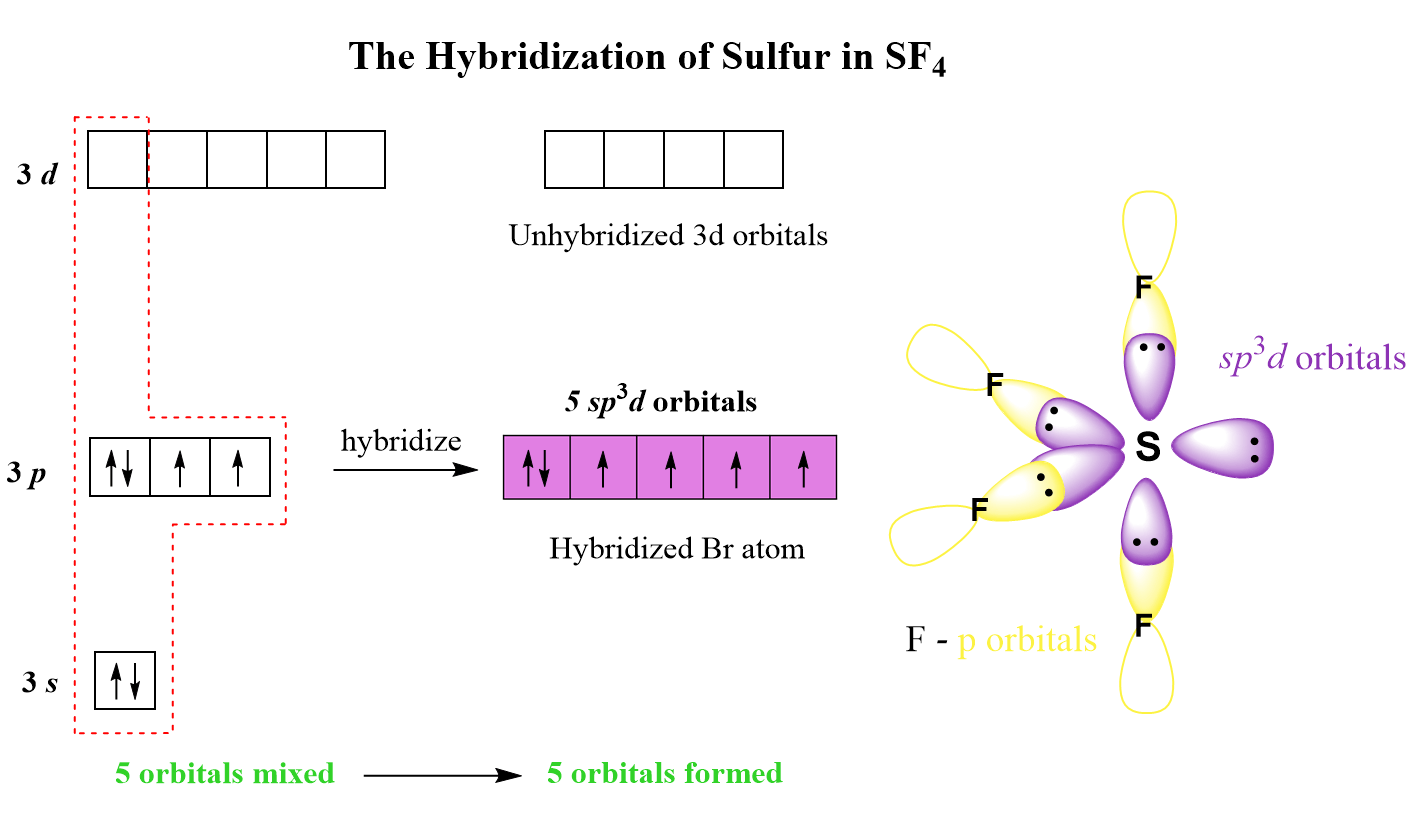
There are 5 units (atoms and lone pairs) on the central atom, and to accommodate them, it needs 5 orbitals which is achieved through sp3d hybridization.
Check this 99-question multiple-choice quiz on Geometry and Hybridization:
Free
Geometry and Hybridization Quiz
Check Also
- The VSEPR Model
- VSEPR Theory Practice Problems
- Hybridization of Atomic Orbitals
- sp, sp2, sp3, sp3d, and sp3d2 Hybridization Practice Problems

